引言
根据联合国艾滋病规划署的《2024年全球艾滋病报告》,全球约4 000万人感染人类免疫缺陷病毒(human immunodeficiency virus,HIV),每年约有130万新的感染者.HIV在感染的早期阶段侵入中枢神经系统,破坏血脑屏障的完整性.血脑屏障功能障碍进一步增强了病毒在中枢神经系统的复制,这导致了HIV相关的脑结构与脑功能损伤.抗逆转录病毒治疗(antiretroviral therapy,ART)是针对HIV的特异性治疗,能够有效抑制HIV复制、改善HIV感染者(people with HIV,PWH)的生活质量,并延长他们的预期寿命[1].尽管严重的HIV相关痴呆症状已被ART有效控制,但近半数接受ART的PWH仍然患有各种与HIV相关的神经系统疾病[2],这可能是由于血脑屏障阻碍了部分抗逆转录病毒药物进入大脑,使得其无法在脑中达到有效浓度,因此HIV在中枢神经系统内维持缓慢且持久的复制[3],另一个原因则是与ART相关的神经和精神不良反应[4].
磁共振成像(magnetic resonance imaging,MRI)是一种强大的非侵入性成像技术[5],已用于探索HIV相关的大脑结构和功能改变[6,7].MRI能够识别ART相关的症状前脑改变,发现与ART相关的脑内正向或负向作用,从而指导辅助治疗.功能磁共振成像(functional MRI,fMRI)基于与神经活动相符的血氧水平变化,可以灵敏地捕捉到神经信号传输的变化.通过fMRI时间序列评估功能连接(functional connectivity,FC)揭示了大脑空间组织的丰富信息[8].传统研究通常假设神经活动保持稳定,FC在扫描过程中保持不变,但事实上,FC具有时间变异性,常规的FC代表了大脑动态活动模式的平均值,一定程度上导致了功能连接的静态连通性在同一疾病的异质性.探究脑中的动态FC(dynamic FC,dFC)有利于更敏锐地发现疾病或药物相关脑改变[9,10].dFC的改变已被报道与帕金森病、阿尔茨海默病等多种神经系统疾病有关[11,12]. 此外,在心理活动不受约束的静息状态下测量得到的血氧水平依赖(blood oxygen level dependent,BOLD)信号已被证明具有内在的时空动态组织[13].
已有大量MRI相关研究表明PWH的大脑结构和功能异常及ART对其的影响,研究发现HIV感染者皮质下区域(杏仁核、尾状核和胼胝体)的脑容量较健康对照组显著减少,而ART未影响大脑结构,且此体积缩小与神经心理测试评分无关[16].这种皮质下萎缩的程度在接受ART的慢性HIV感染者中,与疾病严重程度、年龄和神经认知衰退有关[17].也有文献报道,接受ART的HIV感染者体积减少的区域弥漫地分布在整个大脑.不同方面的神经认知障碍(如图形复制、手指敲击)与不同的灰质区域相关[18].我们之前的研究表明,ART有助于恢复HIV感染者的皮层下-皮层回路的结构连接,且此恢复与免疫水平相关[19]. 此外,静息状态fMRI相关研究表明,12周的ART能有效恢复HIV感染者的平均FC强度及认知评分[20]、微尺度脑动力学和功能网络拓扑指标[21].Ortega等人也发现与未接受治疗的HIV感染者相比,接受ART的HIV感染者的额-纹状体网络有更强的FC[22].增强的FC有时与认知障碍有关,与认知正常的HIV感染者相比,认知障碍的HIV感染者右侧额叶中回与左侧中央后回之间的FC增强[23].此外,与快速ART组(确诊HIV感染后30天内开始ART的参与者)相比,非快速ART组(确诊HIV感染后6个月以上开始ART)的PWH脑灰质体积萎缩和功能改变(包括右侧辅助运动区、左侧枕中回和右侧枕上回等脑区的局部一致性(regional homogeneity,ReHo)明显增加,右脑岛种子区的体素FC强度更弱等)[24].且对未感染HIV的受试者给予两种常见抗逆转录病毒药物:依法韦仑和利托那韦之前和之后,受试者的FC和脑血流量未发生显著变化,一定程度上证明了抗逆转录病毒药物无明显毒性作用[25]. 总之,大多研究表明ART与HIV感染者更好的大脑结构和功能改变有关.但目前还没有研究关注ART对PWH功能连接的动态变化的影响.本研究的目的是调查与ART相关的静息状态功能连接的动态变化,及这些动态变化与临床指标的相关性,从而发现与ART相关的、能敏锐反映恢复或损伤情况的神经成像生物标志物.此外,通过进一步了解在HIV及ART同时作用下PWH脑部的连接模式,有助于提升对ART相关脑功能改变的认识,为辅助治疗提供思路.
1 实验部分
1.1 实验对象
本病例对照研究共纳入了130名PWH和68名健康对照者(healthy control,HC),均为右利手,年龄在23岁至71岁之间.研究程序按照《赫尔辛基宣言》进行,武汉大学中南医院医学伦理委员会批准了本研究(批准号:2011058),并获得了所有参与者的书面知情同意.PWH血清学阳性结果经国家相关实验室确认.根据以下标准排除参与者:(1)有神经系统疾病史、创伤性脑损伤史、药物滥用史或抑郁症史(无排除);(2)疑似并发颅内机会性感染(共排除11名PWH)和脑萎缩(共排除6名PWH);(3)头部过度运动,即平均帧间位移(mean framewise displacement,mean FD)大于0.5 mm(共排除5名PWH).此外,为了使组间性别匹配,将7名接受ART的PWH女性剔除.最终,101名PWH和68名HC被纳入后续分析.在PWH中,45名PWH接受了拉米夫定+齐多夫定/替诺福韦/司他夫定/去羟肌苷+依法韦仑/奈韦拉平/(洛匹那韦/利托那韦)的联合ART超过3个月,56名PWH未接受联合抗逆转录病毒治疗.
1.2 数据采集及预处理
所有MRI数据均在3.0 T西门子Trio扫描仪(Siemens,Erlangen,Germany)和8通道正交鸟笼头线圈上收集.为了减少头部运动和扫描仪噪音的影响,在扫描时使用泡沫垫和耳塞.所有受试者均接受高分辨率3D-T1加权磁化制备的快速采集梯度回波序列,参数为:重复时间(repetition time,TR)/回波时间(echo time,TE)= 1 900 / 2.1 ms;反转时间 = 900 ms;翻转角度 = 9˚;矩阵 = 256 × 256;视场(field of view,FOV)= 240 × 240 mm2;切片厚度为1.0 mm,矢状面切片160片.静息状态fMRI(resting state-fMRI,rs-fMRI)数据采用回波平面成像序列,参数为:TR/TE = 2 000/30 ms;翻转角度 = 90˚;矩阵 = 64 × 64;FOV = 240 × 240 mm2;切片厚度为4.5 mm,轴向切片30片.rs-fMRI扫描持续了420 s,对每个受试者收集210个时间点的扫描.
rs-fMRI数据使用DPABI软件包的rs-fMRI数据处理助手(data processing assistant for resting-state fMRI,DPARSF)协议[26]进行标准化预处理,预处理步骤包括去除前10个时间点、切片时间校正和头动校正.然后将单个3D-T1结构图像与平均功能图像(即所有受试者的rs-fMRI图像的平均图像)共配准,配准后的结构像被分割为灰质、白质和脑脊液:使用统计参数映射(statistical parametric mapping,SPM)工具箱(
1.3 独立成分(independent component,IC)网络提取
数据预处理后,使用fMRI的组独立成分分析工具箱(GICA of fMRI Toolbox,GIFT)[14]中实现的空间GICA框架对对照组和PWH组的功能数据进行分析.首先,采用主成分分析(principal component analysis,PCA)方法,对200个时间点数据进行受试者特定的数据约简步骤,得到150个最大变异的方向;将受试者约简后的数据拼接,并沿最大群体变异方向进一步对该矩阵进行组数据PCA约简,约简为100个分量. 然后,用infomax算法[28]从组主成分约简矩阵中得到100个IC.为了保证估计的稳定性,在ICASSO(
对选出的IC对应的受试者特定时间过程进行去趋势化(消除线性、二次和三次趋势),采用Friston 24个参数的模型[31](包括6个头动参数、1个时间点前的6个头动参数、12个这些参数的平方值)估计受试者的运动参数,并进行回归,以减少头动的影响.然后对其进行去峰抑制,本研究采用功能神经成像分析(analysis of functional neuroimages,AFNI)软件的3DDESPIKE算法进行去峰,此算法基于中位数绝对偏差检测时间过程中的“尖峰”值,并用一个新的数据替换掉峰值,具体信息可参考网址:
1.4 动态功能状态相关分析
采用WBA计算所有IC时间过程两两之间的dFC,窗口大小为22 TRs(44 s),步长为1 TR.窗口形状为锥形,即由高斯卷积(σ =3 TRs)的矩形窗口构成,以获得沿边缘逐渐变细的窗口.并在逆协方差矩阵上施加了一个额外的L1范数约束来加强稀疏性,每个受试者数据得到167个窗.计算完dFC后,使用Fisher的z变换将所有FC矩阵转换为z分数,从而稳定方差以进行进一步分析.
将k-均值聚类算法应用于所有受试者的窗口FC矩阵,以获得跨组重复出现的FC模式(状态),从而进一步评估动态连接模式的频率和结构特性.使用L1距离(曼哈顿距离)函数来估计窗口功能连接矩阵之间的相似性,并利用肘部法则估计最佳的聚类数目为4.此外,在k-均值聚类之前,对每个受试者的时间窗进行子采样,只选择FC方差为局部最大的窗口,把它们作为受试者示例,以减少窗口之间的冗余以及计算需求.
本研究评估了受试者的三项指标,以研究dFC状态的时间特性.包括:(1)每种状态的时间占比(fraction of time,FT),即处于某一状态的时间窗数目;(2)每种状态下的平均停留时间(mean dwell time,MDT),即在切换到另一种状态之前,处于某一状态的连续窗口数的平均值;(3)转换次数(number of transition,NT),以状态从一种状态切换到另一种状态的次数来衡量.
1.5 统计分析
以年龄、性别和mean FD为协变量进行单向协方差分析(analysis of covariance,ANCOVA),以确定存在组间差异的指标.错误发现率(false-discovery rate,FDR)校正用于对差异进行多重比较校正.如果差异显著,则应用独立的双样本t检验或非参数检验来检测每两组指标之间的差异,并使用效应量(Cohen’s d)表示组平均值间的差异程度(计算方式:两组的均值之差除以这两组的合并标准差,0.2~0.3为小效应、0.5左右为中等效应、0.8以上为大效应).并采用偏相关分析方法探讨组间差异显著的动态指标与治疗时间、CD(cluster of differentiation)4计数、蒙特利尔认知评估(montreal cognitive assessment,MoCA)分数之间的关系,回归协变量年龄、性别和mean FD,未进行多重比较校正.显著性水平设为双侧P < 0.05水平.
部分状态的时间特性有显著组间差异,基于网络的统计(network-based statistics,NBS)方法[32,33]被用于定位状态特异性的区域性大脑网络.这是一种能够识别显示某种特定特征的互连网络的方法,本研究利用NBS识别了连接强度有组间差异的互连网络.此方法能够有效地进行多重比较校正,避免过度校正增加假阴性率,且识别出的互连网络能够反映与研究问题相关的连接模式.具体流程如下:首先,为了获得NBS的二值化掩模,对状态的每个功能连接使用以年龄、性别和mean FD为协变量的单向ANCOVA(P < 0.005)来检测三组之间的差异,能够提前限制网络中边的数量.然后利用独立的双尾双样本t检验(P < 0.005),在掩模中确定由连续边组成的相互连接的网络(即图组件),年龄、性别和mean FD作为组间差异的协变量.随后,使用非参数排列方法(排列10 000次),对每个连接组件经验推导出组件大小的零分布,且限制家族误差(family-wise error,FWE)校正后的P < 0.05.连接组件的FWE值通过计算产生相对较大或大小相等的互连网络的排列比例来确定.
2 结果与讨论
2.1 人口统计学与临床资料
最终样本包括45例接受ART治疗的PWH(25位男性,年龄为45.38±10.46岁,mean FD:0.09±0.07 mm),56例未经治疗的PWH(42位男性,年龄为41.07±11.36岁,mean FD:0.010±0.07 mm),68名健康对照者(41位男性,年龄43.34±15.16岁,mean FD:0.09±0.05 mm),三组人口统计学相似(性别:P = 0.09;年龄:P = 0.22;mean FD:P = 0.40).与未经治疗的PWH相比,接受ART治疗的PWH的CD4+ T淋巴细胞更多(P < 0.001).受试者的人口统计学与临床特征见表1.
表1 受试者的人口统计学与临床特征
Table 1
| 未治疗组 | ART治疗组 | 健康对照组 | P值 | |
|---|---|---|---|---|
| 受试者人数 | 56 | 45 | 68 | NA |
| 年龄/岁 | 41.07±11.36 | 45.38±10.46 | 43.34±15.16 | 0.22a |
| 性别(男/女) | 42/14 | 25/20 | 41/27 | 0.09b |
| Mean FD/ mm | 0.10±0.07 | 0.09±0.07 | 0.09±0.05 | 0.40a |
| 诊断时间/月 | 4.45±12.94 | 53.11±35.89 | NA | <0.001c |
| 治疗时间/月 | NA | 41.51±31.70 | NA | NA |
| CD4计数/(cells/mm3) | 116.11±137.67 | 377.05±205.93 | NA | <0.001c |
| MoCA评分 | 23.02±5.53 | 23.40±4.63 | NA | 0.99c |
注:数据以平均值±标准差表示. 诊断时间:确诊至图像扫描时长;NA:不适用或不可用;a: Kruskal-Wallis测试;b: Chi-square测试;c: Mann-Whitney测试.
Note: Data are presented as mean ± standard deviation. NA: not applicable or available; a: Kruskal-Wallis test; b: Chi-square test; c: Mann-Whitney test.
2.2 独立成分网络
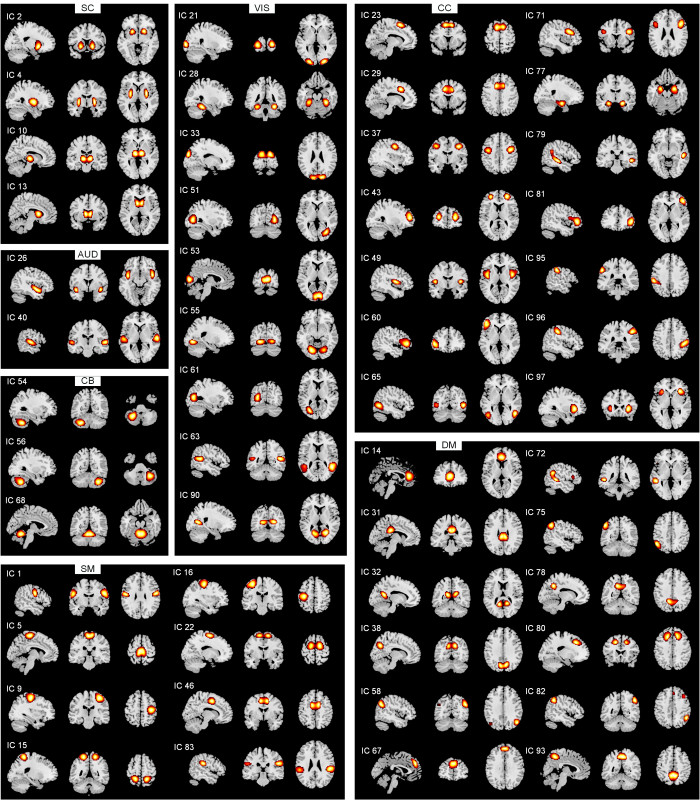
所有52个IC的空间图,使用组ICA识别,详细信息及空间图如图1所示.根据其解剖和功能特性,将独立组件分为以下七个网络:皮层下(subcortical,SC)、听觉(auditory,AUD)、感觉运动(sensorimotor,SM)、视觉(visual,VIS)、认知控制(cognitive control,CC)、默认模式(default mode,DM)和小脑(cerebellum,CB).
图1
图1
7网络中的52个IC的空间图(空间图阈值为t > µ + 5σ)
Fig. 1
Spatial maps for the 52 independent components grouped into seven functional networks (spatial maps were thresholded with t > µ + 5σ)
2.3 动态功能连接状态及分析
2.3.1 时间特性
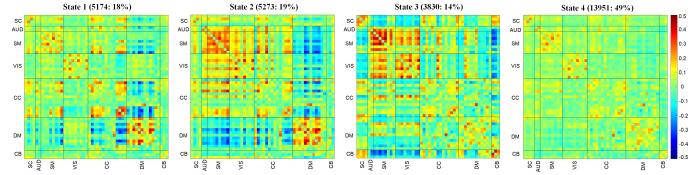
本研究确定了在单个受试者扫描期间、受试者之间反复出现的4种功能连接模式.即,三个频率较低(14%~19%)的状态:各网络间隔离的弱连接状态1,DM网络与其他网络隔离、其他网络整合的状态2,初级网络内部及初级网络间连接最强的状态3;以及频率最高(49%)且连接强度最弱的状态4.图2显示了这4种功能连接状态的可视化连接模式(簇的质心).图2中横、纵坐标表示分属于七个网络中的所有IC;红色表示强连接,绿色表示弱连接,蓝色表示负连接;对角线方框表示网络内部连接,对角线以外为网络间连接.状态1中大部分连接较弱,对角线以外连接均较弱,表示各网络间隔离;状态2中DM网络与其他网络间连接大多为负,表示DM网络与其他网络隔离,而除DM网络外的网络间连接大多为强连接,表示其他网络的整合;状态3中AUD、SM、VIS网络内及网络间连接均为强连接,且这三个网络均为感觉及运动相关的初级网络,表示初级网络的整合状态;状态4中所有连接均为弱连接,且此状态出现频率最高,表示耗能较少的基线状态.对这四个状态的时间特性分别进行组间比较,结果显示仅状态1和状态4的时间特性存在组间差异,因此图3仅展示状态1和状态4的结果.
图2
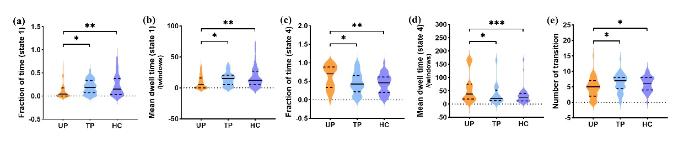
对于状态1,未治疗组(untreated PWH,UP)的FT(P = 0.004,Cohen’s d = 0.570)及MDT(P = 0.004,Cohen’s d = 0.567)显著低于健康对照组(healthy controls,HC),也显著低于ART治疗组(treated PWH,TP)(FT:P = 0.021,Cohen’s d = 0.495; MDT:P = 0.031,Cohen’s d = 0.480);而TP组与HC组无显著差异,如图3(a)、(b).对于状态4,UP组的FT(P = 0.002,Cohen’s d = 0.610)及MDT(P = 0.000 8,Cohen’s d = 0.660)则显著高于HC组,也显著高于TP组(FT:P = 0.012,Cohen’s d = 0.532;MDT:P = 0.027,Cohen’s d = 0.468);而TP组与HC组无显著差异,如图3(c)、(d).此外,相较于UP组,TP组(P = 0.018,Cohen’s d = 0.482)及HC组(P = 0.016,Cohen’s d = 0.454)状态间的NT也显著升高,而TP组与HC组间无显著差异,如图3(e)所示.
图3
图3
时间特性的组间差异. (a)状态1的时间占比. (b)状态1的平均停留时间. (c)状态4的时间占比. (d)状态4的平均停留时间. (e)所有状态间的转换次数. 注:* P<0.05,** P<0.01,*** P<0.001
Fig. 3
Temporal properties differences between groups. (a) Fraction of time in state 1. (b) Mean dwell time in state 1. (c) Fraction of time in state 4. (d) Mean dwell time in state 4. (e) Number of transitions among all states. Note: * P<0.05, ** P<0.01, *** P<0.001
2.3.2 状态网络
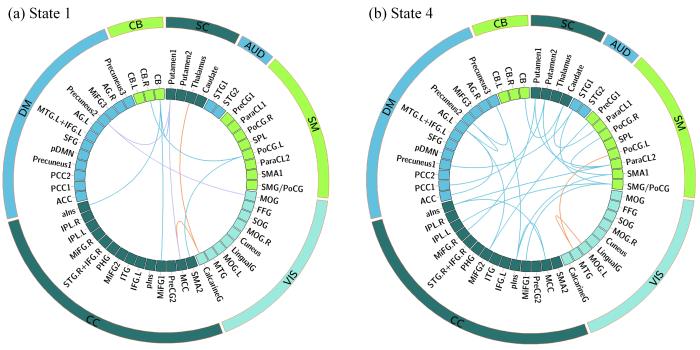
对于时间特性有显著组间差异的状态1、状态4,利用NBS识别出这两个状态具有显著组间差异的连接模式,如图4(a)、(b),其中橙色线条代表TP组与HC组间有显著差异的子网络(P < 0.005),紫色线条代表TP组与UP组间有显著差异的子网络(P < 0.005),蓝色线条代表HC组与UP组间有显著差异的子网络(P < 0.005).状态1的连接模式显示三组间两两差异,TP与HC组间差异的连接涉及SC网络的丘脑(thalamus)与VIS网络的距状回(calcarine gyrus,CalcarineG),及CalcarineG与CC网络的中扣带皮层(middle cingulate cortex,MCC);TP与UP组间差异的连接涉及VIS网络的枕中回(middle occipital gyrus,MOG)、DM网络的楔前叶(precuneus2)、SC网络的壳核(putamen1)及CC网络的MCC;HC与UP组间差异的连接则主要围绕CB网络形成回路,涉及到SM网络的左侧中央后回(left postcentral gyrus,PoCG.L)、CC网络的辅助运动区(supplementary motor area,SMA2)、额中回(middle frontal gyrus,MiFG1)、前脑岛(anterior insula,aIns).状态4的连接模式显示仅HC与TP组间差异(橙色线条)及HC与UP组间差异(蓝色线条),TP与UP组间差异不显著,其中HC与UP的组间差异遍及除VIS网络以外的所有网络,而HC与TP组间差异主要涉及VIS网络的左侧MOG(MOG.L)、颞中回(middle temporal gyrus,MTG),及SM网络的左侧PoCG(PoCG.L).
图4
图4
NBS识别出的显示组间差异的子网络. (a)状态1. (b)状态4. 橙色:TP、HC组间有显著差异的子网络;紫色:TP、UP组间有显著差异的子网络;蓝色:HC、UP组间有显著差异的子网络
Fig. 4
Subnetworks showing differences between groups identified by NBS. (a) State 1. (b) State 4. Orange lines denote subnetwork showing differences between TP and HC; Purple lines denote subnetwork showing differences between TP and UP; Blue lines denote subnetwork showing differences between HC and UP
2.4 临床指标相关性分析
在去除年龄、性别、Mean FD影响后,有组间差异的时间占比、平均停留时间及状态转移次数指标均与治疗时间、CD4计数及MoCA评分没有显著相关性.
2.5 讨论
本研究确定了HIV及ART相关的四种功能连接状态,且从其中状态1及状态4的时间占比、平均停留时间,及所有状态间的转移次数所显示的组间差异不同,表明治疗后明确好转现象.进一步探究这两种状态的连接模式,发现了状态1的小脑相关网络、状态4除视觉网络外的全脑网络显示治疗后好转现象(HC与UP组间差异显著,但TP与HC无组间差异),而状态1、状态4的视觉相关网络均显示治疗后异常现象(TP与HC组间差异显著,但HC与UP无组间差异).此外,状态1中的壳核相关连接则显示治疗后与好转或不良反应均无关的现象.而FC时间指标的改变与治疗时间、CD4计数及MoCA评分无显著相关性.
状态1是除状态4外连接强度最弱的状态,且各网络间隔离、DMN网络与其他网络隔离明显.状态4是所有受试者出现频率最高且连接强度最弱的基线状态.相较于HC组,UP组在状态1的频率明显降低,且停留时间也明显缩短,而在状态4的频率明显升高,且停留时间也明显增加.这表明UP组倾向于异常停留在基线状态,而更少进行网络内部的弱激活,这与HIV相关的FC受损有关[34].而TP组的这些指标均与HC组差异不明显,这表明TP组由于HIV被抑制,脑FC动态特性也有效恢复,相关研究也证明了此观点[20].此外,UP组的状态转移次数较另外两组也有明显降低,这表明HIV影响了UP组的动态灵活性,而ART使灵活性有效恢复[21].
由NBS识别出的状态1与状态4的组间差异连接模式中,ART相关的不良反应均表现在视觉相关的连接,这可能是由于抗逆转录病毒的药物易损伤视觉相关脑区[35],从而影响视觉相关回路的功能.ART相关的治疗后好转在状态1中主要表现在小脑相关的连接,小脑作为重要的运动协调区域,其与大脑皮层脑区间的传入和传出连接的恢复有助于恢复机体平衡及运动等功能、以及改善认知功能[36].ART相关的治疗反应在状态1则主要表现在皮层下(壳核)相关连接,本文作者之前的研究结果表明壳核在MRI结构像中显示明确的治疗后好转[19],这可能是由于患病机体为弥补结构损伤而进行的功能代偿,从而使得其与健康对照组差异不明显.ART相关的治疗后好转在状态4中表现在除视觉网络外的广泛的网络连接中,状态4是所有受试者中出现频率最高的基线状态,这表现了ART能有效恢复HIV感染者的网络连接.改变指标的临床相关性弱可能是由于功能改变常常发生于临床指标发生大幅波动之前.
此外,本研究具有一定的局限性:由于本研究是横断面研究,缺乏治疗组在治疗前的基线状态,无法确定是否是由于基线生理数据的差异导致治疗组与未治疗组的脑功能差异.因此研究结论需要纵向队列研究进一步验证,但本研究结果可为HIV相关脑功能异常的队列研究及实验性研究提供病因研究的线索和方向.
3 结论
本研究通过研究接受及未接受ART的PWH静息状态FC的动态特性,发现了未治疗的PWH倾向于异常停留在基线状态,而更少停留在网络内部的弱激活状态,且转换状态更不灵活,而ART有助于恢复PWH的动态特性.此外,在网络内部的弱激活状态,ART恢复体现在小脑相关连接,而壳核可能与功能代偿有关;在基线状态,ART恢复则体现在除视觉网络外的广泛网络连接.视觉相关连接在两种状态中均体现为ART相关的不良反应.
利益冲突
无
参考文献
Immune recovery in HIV-1 infected patients with sustained viral suppression under long-term antiretroviral therapy in Ethiopia
[J].DOI:10.1371/journal.pone.0240880 URL [本文引用: 1]
Prevalence and 1-year incidence of HIV-associated neurocognitive disorder (HAND) in adults aged ≥50 years attending standard HIV clinical care in Kilimanjaro, Tanzania
[J].DOI:10.1017/S1041610221000156 URL [本文引用: 1]
The Paradox of HIV blood-brain barrier penetrance and antiretroviral drug delivery deficiencies
[J].
DOI:S0166-2236(20)30149-1
PMID:32682564
[本文引用: 1]

HIV attacks the body's immune cells, frequently compromises the integrity of the blood-brain barrier (BBB), and infects the CNS in the early stages of infection. Dysfunction of the BBB further potentiates viral replication within the CNS, which can lead to HIV-associated neuropathology. Antiretroviral therapy (ART) significantly improves HIV patient outcomes and reduces mortality rates. However, there has been limited progress in targeting latent viral reservoirs within the CNS, which may eventually lead to rebound viremia. While ART drugs are shown to be effective in attenuating HIV replication in the periphery, the protection of the brain by the BBB offers an isolated sanctuary to harbor HIV and maintains chronic and persistent replication within the CNS. In this review, we elucidate the pathology of the BBB, its ability to potentiate viral replication, as well as current therapies and insufficiencies in treating HIV-infected individuals.Copyright © 2020 The Author(s). Published by Elsevier Ltd.. All rights reserved.
Nucleoside reverse transcriptase inhibitors (NRTIs) induce proinflammatory cytokines in the CNS via Wnt5a signaling
[J].
DOI:10.1038/s41598-017-03446-w
PMID:28646196
[本文引用: 1]

HAART is very effective in suppressing HIV-1 replication in patients. However, patients staying on long-term HAART still develop various HIV-associated neurological disorders, even when the viral load is low. The underlying pathogenic mechanisms are largely unknown. Emerging evidence implicated that persistent neuroinflammation plays an important role in NeuroAIDS. Although residual virus or viral proteins are commonly thought as the causal factors, we are interested in the alternative possibility that HAART critically contributes to the neuroinflammation in the central nervous system (CNS). To test this hypothesis, we have determined the effect of NRTIs on the expression of proinflammatory cytokines in the various CNS regions. Mice (C57Bl/6) were administered with AZT (Zidovudine 100 mg/kg/day), 3TC (Lamivudine 50 mg/kg/day) or D4T (Stavudine 10 mg/kg/day) for 5 days, and cortices, hippocampi and spinal cords were collected for immunoblotting. Our results showed that NRTI administration up-regulated cytokines, including IL-1β, TNF-α and IL-6 in various CNS regions. In addition, we found that NRTIs also up-regulated Wnt5a protein. Importantly, BOX5 attenuated NRTI-induced cytokine up-regulation. These results together suggest that NRTIs up-regulate proinflammatory cytokines via a Wnt5a signaling-dependent mechanism. Our findings may help understand the potential pathogenic mechanisms of HAART-associated NeuroAIDS and design effective adjuvants.
Application of magnetic resonance imaging technology in pediatric exercise intervention research
[J].
磁共振成像技术在儿童运动干预研究中的应用
[J].
DOI:10.11938/cjmr20243129
[本文引用: 1]

运动干预作为辅助儿童疾病康复和促进儿童大脑结构功能改善的方法备受关注.磁共振成像技术提供了多种分析方法研究儿童大脑变化,在儿童运动干预研究中得到广泛应用.本文通过分析现有的儿童运动干预研究成果,进一步深入探讨运动干预对儿童大脑的影响、研究中影像技术和干预方法的选择,分析了部分实验差异不明显的原因,并提出了解决建议.本综述总结了磁共振成像技术在儿童运动干预研究中的应用,分析了其重要作用及潜在价值,为后续相关研究提供了有益的参考.
Brain structural changes following HIV infection: meta-analysis
[J].DOI:10.3174/ajnr.A5432 URL [本文引用: 1]
HIV infection and the fronto-striatal system: a systematic review and meta-analysis of fMRI studies
[J].
DOI:10.1097/QAD.0000000000000151
PMID:24300546
[本文引用: 1]

Functional MRI studies investigating the impact of HIV on the brain have implicated the involvement of fronto-striatal circuitry. However, to date there is no review and meta-analysis of this work. We systematically reviewed the literature and performed a meta-analysis of functional magnetic resonance imaging (fMRI) studies in HIV-infected individuals using a well validated tool recently developed for use in fMRI, 'GingerALE'. Twenty-one studies (468 HIV+, 270 HIV- controls) were qualitatively reviewed, of which six (105 HIV+, 102 controls) utilized fMRI paradigms engaging the fronto-striatal-parietal network, making a quantitative analysis possible. Our meta-analysis revealed consistent functional differences in the left inferior frontal gyrus and caudate nucleus between infected participants and controls across these studies. This fronto-striatal dysfunction was qualitatively related to cognitive impairment, disease progression and treatment effects. Although further work needs to be done to further delineate the potentially confounding influence of substance abuse and HIV-related comorbidities, as well as HIV's effect on functional haemodynamic vascular coupling, these findings indicate that further investigation of the fronto-striatal sub-networks in HIV-infected patients is warranted.
Three types of individual variation in brain networks revealed by single-subject functional connectivity analyses
[J].
Tools of the trade: estimating time-varying connectivity patterns from fMRI data
[J].
DOI:10.1093/scan/nsaa114
URL
[本文引用: 1]

Given the dynamic nature of the brain, there has always been a motivation to move beyond ‘static’ functional connectivity, which characterizes functional interactions over an extended period of time. Progress in data acquisition and advances in analytical neuroimaging methods now allow us to assess the whole brain’s dynamic functional connectivity (dFC) and its network-based analog, dynamic functional network connectivity at the macroscale (mm) using fMRI. This has resulted in the rapid growth of analytical approaches, some of which are very complex, requiring technical expertise that could daunt researchers and neuroscientists. Meanwhile, making real progress toward understanding the association between brain dynamism and brain disorders can only be achieved through research conducted by domain experts, such as neuroscientists and psychiatrists. This article aims to provide a gentle introduction to the application of dFC. We first explain what dFC is and the circumstances under which it can be used. Next, we review two major categories of analytical approaches to capture dFC. We discuss caveats and considerations in dFC analysis. Finally, we walk readers through an openly accessible toolbox to capture dFC properties and briefly review some of the dynamic metrics calculated using this toolbox.
Time-varying analysis of brain networks based on high-order dynamic functional connections in mild cognitive impairment
[J].
基于轻度认知障碍高阶动态功能连接的脑网络时变分析
[J].
DOI:10.11938/cjmr20243102
[本文引用: 1]

现有研究常用功能连接(FC)结合图论分析完成轻度认知障碍(MCI)疾病的辅助诊断.传统FC分析方法通常以低阶FC网络为对象,而高阶FC网络能够揭示脑网络中更高层次的交互关系,但在高阶FC网络中涉及图论研究尚少,且传统图论指标在高阶FC网络中具有局限性.本文通过高阶动态功能连接构建高阶FC网络,结合图论对MCI和正常认知(NC)的脑网络状态进行分析,定义了阻滞系数和平均转换时间两个新的图论指标,以表征脑网络的时间变异性.结果表明在高阶FC网络中应用图论能有效提取MCI组和NC组之间的差异性信息,所提出的阻滞系数和平均转换时间指标均能呈现显著性差异,为高阶脑网络的研究提供了一种新的分析方法.
Abnormal dynamic functional connectivity in Alzheimer’s disease
[J].DOI:10.1111/cns.v26.9 URL [本文引用: 1]
Abnormal intrinsic brain functional network dynamics in Parkinson’s disease
[J].DOI:10.1093/brain/awx233 URL [本文引用: 1]
Time-varying functional network information extracted from brief instances of spontaneous brain activity
[J].
DOI:10.1073/pnas.1216856110
PMID:23440216
[本文引用: 1]

Recent functional magnetic resonance imaging studies have shown that the brain is remarkably active even in the absence of overt behavior, and this activity occurs in spatial patterns that are reproducible across subjects and follow the brain's established functional subdivision. Investigating the distribution of these spatial patterns is an active area of research with the goal of obtaining a better understanding of the neural networks underlying brain function. One intriguing aspect of spontaneous activity is an apparent nonstationarity, or variability of interaction between brain regions. It was recently proposed that spontaneous brain activity may be dominated by brief traces of activity, possibly originating from a neuronal avalanching phenomenon. Such traces may involve different subregions in a network at different times, potentially reflecting functionally relevant relationships that are not captured with conventional data analysis. To investigate this, we examined publicly available functional magnetic resonance imaging data with a dedicated analysis method and found indications that functional networks inferred from conventional correlation analysis may indeed be driven by activity at only a few critical time points. Subsequent analysis of the activity at these critical time points revealed multiple spatial patterns, each distinctly different from the established functional networks. The spatial distribution of these patterns suggests a potential functional relevance.
A method for making group inferences from functional MRI data using independent component analysis
[J].
DOI:10.1002/hbm.1048
PMID:11559959
[本文引用: 3]

Independent component analysis (ICA) is a promising analysis method that is being increasingly applied to fMRI data. A principal advantage of this approach is its applicability to cognitive paradigms for which detailed models of brain activity are not available. Independent component analysis has been successfully utilized to analyze single-subject fMRI data sets, and an extension of this work would be to provide for group inferences. However, unlike univariate methods (e.g., regression analysis, Kolmogorov-Smirnov statistics), ICA does not naturally generalize to a method suitable for drawing inferences about groups of subjects. We introduce a novel approach for drawing group inferences using ICA of fMRI data, and present its application to a simple visual paradigm that alternately stimulates the left or right visual field. Our group ICA analysis revealed task-related components in left and right visual cortex, a transiently task-related component in bilateral occipital/parietal cortex, and a non-task-related component in bilateral visual association cortex. We address issues involved in the use of ICA as an fMRI analysis method such as: (1) How many components should be calculated? (2) How are these components to be combined across subjects? (3) How should the final results be thresholded and/or presented? We show that the methodology we present provides answers to these questions and lay out a process for making group inferences from fMRI data using independent component analysis.Copyright 2001 Wiley-Liss, Inc.
Dynamic functional connectivity: Promise, issues, and interpretations
[J].
DOI:10.1016/j.neuroimage.2013.05.079
PMID:23707587
[本文引用: 1]

The brain must dynamically integrate, coordinate, and respond to internal and external stimuli across multiple time scales. Non-invasive measurements of brain activity with fMRI have greatly advanced our understanding of the large-scale functional organization supporting these fundamental features of brain function. Conclusions from previous resting-state fMRI investigations were based upon static descriptions of functional connectivity (FC), and only recently studies have begun to capitalize on the wealth of information contained within the temporal features of spontaneous BOLD FC. Emerging evidence suggests that dynamic FC metrics may index changes in macroscopic neural activity patterns underlying critical aspects of cognition and behavior, though limitations with regard to analysis and interpretation remain. Here, we review recent findings, methodological considerations, neural and behavioral correlates, and future directions in the emerging field of dynamic FC investigations. Copyright © 2013 Elsevier Inc. All rights reserved.
Independent effects of HIV, aging, and HAART on brain volumetric measures
[J].
DOI:10.1097/QAI.0b013e318249db17
PMID:22269799
[本文引用: 1]

Neurocognitive impairment remains prevalent in HIV-infected (HIV+) individuals despite highly active antiretroviral therapy (HAART). We assessed the impact of HIV, HAART, and aging using structural neuroimaging.Seventy-eight participants [HIV- (n = 26), HIV+ on stable HAART (HIV+/HAART+; n = 26), HIV+ naive to HAART (HIV+/HAART-; n = 26)] completed neuroimaging and neuropsychological testing. A subset of HIV+ subjects (n = 12) performed longitudinal assessments before and after initiating HAART. Neuropsychological tests evaluated memory, psychomotor speed, and executive function, and a composite neuropsychological score was calculated based on normalized performances (neuropsychological summary Z score, NPZ-4). Volumetrics were evaluated for the amygdala, caudate, thalamus, hippocampus, putamen, corpus callosum, and cerebral gray and white matter. A 3-group 1-way analysis of variance assessed differences in neuroimaging and neuropsychological indices. Correlations were examined between NPZ-4 and volumetrics. Exploratory testing using a broken-stick regression model evaluated self-reported duration of HIV infection on brain structure.HIV+ individuals had significant reductions in brain volumetrics within select subcortical regions (amygdala, caudate, and corpus callosum) compared with HIV- participants. However, HAART did not affect brain structure as regional volumes were similar for HIV+/HAART- and HIV+/HAART+. No association existed between NPZ-4 and volumetrics. HIV and aging were independently associated with volumetric reductions. Exploratory analyses suggest caudate atrophy due to HIV slowly occurs after self-reported seroconversion.HIV associated volumetric reductions within the amygdala, caudate, and corpus callosum occurs despite HAART. A gradual decline in caudate volume occurs after self-reported seroconversion. HIV and aging independently increase brain vulnerability. Additional longitudinal structural magnetic resonance imaging studies, especially within older HIV+ participants, are required.
Progressive brain atrophy in chronically infected and treated HIV+ individuals
[J].
DOI:10.1007/s13365-019-00723-4
PMID:30767174
[本文引用: 1]

Growing evidence points to persistent neurological injury in chronic HIV infection. It remains unclear whether chronically HIV-infected individuals on combined antiretroviral therapy (cART) develop progressive brain injury and impaired neurocognitive function despite successful viral suppression and immunological restoration. In a longitudinal neuroimaging study for the HIV Neuroimaging Consortium (HIVNC), we used tensor-based morphometry to map the annual rate of change of regional brain volumes (mean time interval 1.0 ± 0.5 yrs), in 155 chronically infected and treated HIV+ participants (mean age 48.0 ± 8.9 years; 83.9% male). We tested for associations between rates of brain tissue loss and clinical measures of infection severity (nadir or baseline CD4+ cell count and baseline HIV plasma RNA concentration), HIV duration, cART CNS penetration-effectiveness scores, age, as well as change in AIDS Dementia Complex stage. We found significant brain tissue loss across HIV+ participants, including those neuro-asymptomatic with undetectable viral loads, largely localized to subcortical regions. Measures of disease severity, age, and neurocognitive decline were associated with greater atrophy. Chronically HIV-infected and treated individuals may undergo progressive brain tissue loss despite stable and effective cART, which may contribute to neurocognitive decline. Understanding neurological complications of chronic infection and identifying factors associated with atrophy may help inform strategies to maintain brain health in people living with HIV.
Neurocognitive impairment and gray matter volume reduction in HIV-infected patients
[J].
DOI:10.1007/s13365-020-00865-w
PMID:32572834
[本文引用: 1]

Although neuropsychological studies of human immunodeficiency virus (HIV)-infected patients have demonstrated heterogeneity in neurocognitive impairment and neuroimaging studies have reported diverse brain regions affected by HIV, it remains unclear whether individual differences in neurocognitive impairment are underpinned by their neural bases. Here, we investigated spatial distribution patterns of correlation between neurocognitive function and regional gray matter (GM) volume across patients with HIV. Thirty-one combination antiretroviral therapy-treated HIV-infected Japanese male patients and 33 age- and sex-matched healthy controls were included in the analysis after strict exclusion criteria, especially for substance use. Fifteen neurocognitive tests were used, and volumetric magnetic resonance imaging was performed. We used voxel-based morphometry to compare GM volume between groups and identify regional GM volumes that correlated with neurocognitive tests across patients. Using the Frascati criteria, 10 patients were diagnosed with asymptomatic neurocognitive impairment, while the others were not diagnosed with HIV-associated neurocognitive disorders. Patients showed a significantly lower performance in five neurocognitive tests as well as significantly reduced GM volume relative to controls, with volume-reduced regions spread diffusely across the whole brain. Different aspects of neurocognitive impairment (i.e., figural copy, finger tapping, and Pegboard) were associated with different GM regions. Our findings suggest a biological background constituting heterogeneity of neurocognitive impairment in HIV infection and support the clinical importance of considering individual differences for tailor-made medicine for people living with HIV.
Highly active antiretroviral therapy-related effects on morphological connectivity in HIV
[J].
DOI:10.1097/QAD.0000000000003759
URL
[本文引用: 2]

Suboptimal concentration of the antiretroviral drug is insufficient to inhibit HIV destruction on brain structure and function due to the resistance of blood brain barrier. We aimed to investigate highly active antiretroviral therapy (HAART)-related effects on the morphological connectivity in people with HIV (PWH).
Combination antiretroviral therapy improves cognitive performance and functional connectivity in treatment-naïve HIV-infected individuals
[J].
DOI:10.1007/s13365-017-0553-9
PMID:28791662
[本文引用: 2]

Our study aimed to investigate the short-term effect of combination antiretroviral therapy (cART) on cognitive performance and functional and structural connectivity and their relationship to plasma levels of antiretroviral (ARV) drugs. Seventeen ARV treatment-naïve HIV-infected individuals (baseline mean CD4 cell count, 479 ± 48 cells/mm) were age matched with 17 HIV-uninfected individuals. All subjects underwent a detailed neurocognitive and functional assessment and magnetic resonance imaging. HIV-infected subjects were scanned before starting cART and 12 weeks after initiation of treatment. Uninfected subjects were assessed once at baseline. Functional connectivity (FC) was assessed within the default mode network while structural connectivity was assessed by voxel-wise analysis using tract-based spatial statistics (TBSS) and probabilistic tractography within the DMN. Tenofovir and emtricitabine blood concentration were measured at week 12 of cART. Prior to cART, HIV-infected individuals had significantly lower cognitive performance than control subjects as measured by the total Z-score from the neuropsychological tests assessing six cognitive domains (p = 0.020). After 12 weeks of cART treatment, there remained only a weak cognitive difference between HIV-infected and HIV-uninfected subjects (p = 0.057). Mean FC was lower in HIV-infected individuals compared with those uninfected (p = 0.008), but FC differences became non-significant after treatment (p = 0.197). There were no differences in DTI metrics between HIV-infected and HIV-uninfected individuals using the TBSS approach and limited evidence of decreased structural connectivity within the DMN in HIV-infected individuals. Tenofovir and emtricitabine plasma concentrations did not correlate with either cognitive performance or imaging metrics.Twelve weeks of cART improves cognitive performance and functional connectivity in ARV treatment-naïve HIV-infected individuals with relatively preserved immune function. Longer periods of observation are necessary to assess whether this effect is maintained.
Whole-brain computational modeling reveals disruption of microscale brain dynamics in HIV infected individuals
[J].DOI:10.1002/hbm.v42.1 URL [本文引用: 2]
Effects of HIV and combination antiretroviral therapy on cortico-striatal functional connectivity
[J].
DOI:10.1097/QAD.0000000000000611
PMID:25849834
[本文引用: 1]

Determine whether HIV and combination antiretroviral therapy (cART) affect resting-state functional connectivity (rs-fc) between the striatum and the cortical regions.Forty-nine HIV-uninfected (HIV-) and 132 HIV-infected (HIV+) (65% receiving cART) patients underwent laboratory studies (current and nadir CD4 T-cell counts, and plasma HIV viral load), neuropsychological performance testing, and neuroimaging. Rs-fc, which examines the coordination of neural activity in distant brain regions, was used to investigate the cortico-striatal functional connections. The effect of cART was assessed comparing HIV+ individuals on cART (HIV+/cART+), and HIV+ individuals not currently receiving cART (HIV+/cART-). Relationships between laboratory tests, cognitive performance, and cART on subcortical-cortical rs-fc were assessed by an analysis of variance.HIV+ individuals had lower cortico-striatal functional connectivity than HIV- controls, specifically between the striatum and the default mode network (P < 0.001) and ventral attention network (P < 0.001). HIV+/cART+ individuals had higher functional connectivity between the striatum, and default mode network (P = 0.02) and ventral attention network (P = 0.01), compared to the HIV+/cART- patients. Laboratory (current and nadir CD4 T-cell counts, plasma viral load) and neuropsychological performance was not correlated with cortico-striatal rs-fc.HIV was associated with disrupted cortico-striatal networks, consistent with HIV's known impact on the subcortical areas. Interestingly, within certain networks, HIV+/cART+ individuals had similar rs-fc compared to HIV- controls, suggesting possible improvements in HIV-related neural dysfunction due to medications. Rs-fc may be a sensitive biomarker of neural insult and its recovery following cART. Additional studies may show rs-fc has utility in measuring acute inflammation caused by HIV.
CD8+T cells and monocytes were associated with brain alterations in human immunodeficiency virus-infected individuals with cognitive impairment
[J].DOI:10.1016/j.brainresbull.2025.111231 URL [本文引用: 1]
The association between rapid antiretroviral therapy initiation and brain structure and function based on multimodal magnetic resonance imaging in HIV-positive men who have sex with men
[J].
DOI:10.1186/s12879-024-10397-x
PMID:39780061
[本文引用: 1]

An increasing number of treatment guidelines recommend rapid initiation of antiretroviral therapy (ART) after the diagnosis of human immunodeficiency virus (HIV) infection. However, data on the association between rapid ART initiation and alterations in brain structure and function remain limited in people with HIV (PWH). A cross-sectional analysis was conducted on HIV-positive men who have sex with men (MSM) undergoing ART. Fifty-four participants who started ART within 30 days of confirmed HIV diagnosis (rapid ART group) and 20 participants who started ART more than 6 months of confirmed HIV diagnosis (non-rapid ART group) completed clinical assessments and multimodal magnetic resonance imaging scans to obtain both anatomical and resting-state functional images. Compared to PWH in the non-rapid ART group, those in the rapid ART group exhibited a greater total gray matter volume (P = 0.001) and functional changes, including a lower amplitude of low-frequency fluctuations in the left angular gyrus (P < 0.001). Moreover, the results of the main effects and interactions indicated that rapid ART initiation had main effects on major imaging outcomes. The validation analysis results in participants who started ART within 7 days of confirmed HIV diagnosis generally corroborated and complemented the aforementioned findings. Our study demonstrated brain gray matter volume atrophy and functional alterations in PWH of the non-rapid ART group compared to those in the rapid ART group, suggesting that rapid ART initiation may be associated with better brain structure and function changes in HIV-positive MSM.© 2025. The Author(s).
Effect of HAART on brain organization and function in HIV-negative subjects
[J].DOI:10.1007/s11481-015-9634-9 URL [本文引用: 1]
DPARSF: a MATLAB toolbox for "pipeline" data analysis of resting-state fMRI
[J].
A fast diffeomorphic image registration algorithm
[J].
DOI:10.1016/j.neuroimage.2007.07.007
PMID:17761438
[本文引用: 1]

This paper describes DARTEL, which is an algorithm for diffeomorphic image registration. It is implemented for both 2D and 3D image registration and has been formulated to include an option for estimating inverse consistent deformations. Nonlinear registration is considered as a local optimisation problem, which is solved using a Levenberg-Marquardt strategy. The necessary matrix solutions are obtained in reasonable time using a multigrid method. A constant Eulerian velocity framework is used, which allows a rapid scaling and squaring method to be used in the computations. DARTEL has been applied to intersubject registration of 471 whole brain images, and the resulting deformations were evaluated in terms of how well they encode the shape information necessary to separate male and female subjects and to predict the ages of the subjects.
An information-maximization approach to blind separation and blind deconvolution
[J].
DOI:10.1162/neco.1995.7.6.1129
PMID:7584893
[本文引用: 1]

We derive a new self-organizing learning algorithm that maximizes the information transferred in a network of nonlinear units. The algorithm does not assume any knowledge of the input distributions, and is defined here for the zero-noise limit. Under these conditions, information maximization has extra properties not found in the linear case (Linsker 1989). The nonlinearities in the transfer function are able to pick up higher-order moments of the input distributions and perform something akin to true redundancy reduction between units in the output representation. This enables the network to separate statistically independent components in the inputs: a higher-order generalization of principal components analysis. We apply the network to the source separation (or cocktail party) problem, successfully separating unknown mixtures of up to 10 speakers. We also show that a variant on the network architecture is able to perform blind deconvolution (cancellation of unknown echoes and reverberation in a speech signal). Finally, we derive dependencies of information transfer on time delays. We suggest that information maximization provides a unifying framework for problems in "blind" signal processing.
A baseline for the multivariate comparison of resting-state networks
[J].
DOI:10.3389/fnsys.2011.00002
PMID:21442040
[本文引用: 1]

As the size of functional and structural MRI datasets expands, it becomes increasingly important to establish a baseline from which diagnostic relevance may be determined, a processing strategy that efficiently prepares data for analysis, and a statistical approach that identifies important effects in a manner that is both robust and reproducible. In this paper, we introduce a multivariate analytic approach that optimizes sensitivity and reduces unnecessary testing. We demonstrate the utility of this mega-analytic approach by identifying the effects of age and gender on the resting-state networks (RSNs) of 603 healthy adolescents and adults (mean age: 23.4 years, range: 12-71 years). Data were collected on the same scanner, preprocessed using an automated analysis pipeline based in SPM, and studied using group independent component analysis. RSNs were identified and evaluated in terms of three primary outcome measures: time course spectral power, spatial map intensity, and functional network connectivity. Results revealed robust effects of age on all three outcome measures, largely indicating decreases in network coherence and connectivity with increasing age. Gender effects were of smaller magnitude but suggested stronger intra-network connectivity in females and more inter-network connectivity in males, particularly with regard to sensorimotor networks. These findings, along with the analysis approach and statistical framework described here, provide a useful baseline for future investigations of brain networks in health and disease.
Tracking whole-brain connectivity dynamics in the resting state
[J].
DOI:10.1093/cercor/bhs352
PMID:23146964
[本文引用: 1]

Spontaneous fluctuations are a hallmark of recordings of neural signals, emergent over time scales spanning milliseconds and tens of minutes. However, investigations of intrinsic brain organization based on resting-state functional magnetic resonance imaging have largely not taken into account the presence and potential of temporal variability, as most current approaches to examine functional connectivity (FC) implicitly assume that relationships are constant throughout the length of the recording. In this work, we describe an approach to assess whole-brain FC dynamics based on spatial independent component analysis, sliding time window correlation, and k-means clustering of windowed correlation matrices. The method is applied to resting-state data from a large sample (n = 405) of young adults. Our analysis of FC variability highlights particularly flexible connections between regions in lateral parietal and cingulate cortex, and argues against a labeling scheme where such regions are treated as separate and antagonistic entities. Additionally, clustering analysis reveals unanticipated FC states that in part diverge strongly from stationary connectivity patterns and challenge current descriptions of interactions between large-scale networks. Temporal trends in the occurrence of different FC states motivate theories regarding their functional roles and relationships with vigilance/arousal. Overall, we suggest that the study of time-varying aspects of FC can unveil flexibility in the functional coordination between different neural systems, and that the exploitation of these dynamics in further investigations may improve our understanding of behavioral shifts and adaptive processes.
Movement-related effects in fMRI time-series
[J].
DOI:10.1002/mrm.1910350312
PMID:8699946
[本文引用: 1]

This paper concerns the spatial and intensity transformations that are required to adjust for the confounding effects of subject movement during functional MRI (fMRI) activation studies. An approach is presented that models, and removes, movement-related artifacts from fMRI time-series. This approach is predicated on the observation that movement-related effects are extant even after perfect realignment. Movement-related effects can be divided into those that are a function of position of the object in the frame of reference of the scanner and those that are due to movement in previous scans. This second component depends on the history of excitation experienced by spins in a small volume and consequent differences in local saturation. The spin excitation history thus will itself be a function of previous positions, suggesting an autoregression-moving average model for the effects of previous displacements on the current signal. A model is described as well as the adjustments for movement-related components that ensue. The empirical analyses suggest that (in extreme situations) over 90% of fMRI signal can be attributed to movement, and that this artifactual component can be successfully removed.
Network-based statistic: Identifying differences in brain networks
[J].
DOI:10.1016/j.neuroimage.2010.06.041
PMID:20600983
[本文引用: 1]

Large-scale functional or structural brain connectivity can be modeled as a network, or graph. This paper presents a statistical approach to identify connections in such a graph that may be associated with a diagnostic status in case-control studies, changing psychological contexts in task-based studies, or correlations with various cognitive and behavioral measures. The new approach, called the network-based statistic (NBS), is a method to control the family-wise error rate (in the weak sense) when mass-univariate testing is performed at every connection comprising the graph. To potentially offer a substantial gain in power, the NBS exploits the extent to which the connections comprising the contrast or effect of interest are interconnected. The NBS is based on the principles underpinning traditional cluster-based thresholding of statistical parametric maps. The purpose of this paper is to: (i) introduce the NBS for the first time; (ii) evaluate its power with the use of receiver operating characteristic (ROC) curves; and, (iii) demonstrate its utility with application to a real case-control study involving a group of people with schizophrenia for which resting-state functional MRI data were acquired. The NBS identified a expansive dysconnected subnetwork in the group with schizophrenia, primarily comprising fronto-temporal and occipito-temporal dysconnections, whereas a mass-univariate analysis controlled with the false discovery rate failed to identify a subnetwork.Copyright © 2010 Elsevier Inc. All rights reserved.
GRETNA: a graph theoretical network analysis toolbox for imaging connectomics
[J].
DOI:10.3389/fnhum.2015.00386
PMID:26175682
[本文引用: 1]

Recent studies have suggested that the brain's structural and functional networks (i.e., connectomics) can be constructed by various imaging technologies (e.g., EEG/MEG; structural, diffusion and functional MRI) and further characterized by graph theory. Given the huge complexity of network construction, analysis and statistics, toolboxes incorporating these functions are largely lacking. Here, we developed the GRaph thEoreTical Network Analysis (GRETNA) toolbox for imaging connectomics. The GRETNA contains several key features as follows: (i) an open-source, Matlab-based, cross-platform (Windows and UNIX OS) package with a graphical user interface (GUI); (ii) allowing topological analyses of global and local network properties with parallel computing ability, independent of imaging modality and species; (iii) providing flexible manipulations in several key steps during network construction and analysis, which include network node definition, network connectivity processing, network type selection and choice of thresholding procedure; (iv) allowing statistical comparisons of global, nodal and connectional network metrics and assessments of relationship between these network metrics and clinical or behavioral variables of interest; and (v) including functionality in image preprocessing and network construction based on resting state functional MRI (R-fMRI) data. After applying the GRETNA to a publicly released R-fMRI dataset of 54 healthy young adults, we demonstrated that human brain functional networks exhibit efficient small-world, assortative, hierarchical and modular organizations and possess highly connected hubs and that these findings are robust against different analytical strategies. With these efforts, we anticipate that GRETNA will accelerate imaging connectomics in an easy, quick and flexible manner. GRETNA is freely available on the NITRC website.
Pathways to neurodegeneration
[J].DOI:10.1212/WNL.0b013e318288792b URL [本文引用: 1]
The neuro-ophthalmology of HIV-AIDS review of neurobehavioral HIV medicine
[J].
Cerebellar-subcortical-cortical systems as modulators of cognitive functions
[J].
DOI:10.1007/s11065-020-09465-1
PMID:33515170
[本文引用: 1]

Over the past few decades, research has established that the cerebellum is involved in executive functions; however, its specific role remains unclear. There are numerous theories of cerebellar function and numerous cognitive processes falling under the umbrella of executive function, making investigations of the cerebellum's role in executive functioning challenging. In this review, we explored the role of the cerebellum in executive functioning through clinical and cognitive neuroscience frameworks. We reviewed the neuroanatomical systems and theoretical models of cerebellar functions and the multifaceted nature of executive functions. Using attention deficit hyperactivity disorder and cerebellar tumor as clinical developmental models of cerebellar dysfunction, and the functional magnetic resonance imaging literature, we reviewed evidence for cerebellar involvement in specific components of executive function in childhood, adolescence, and adulthood. There is evidence for posterior cerebellar contributions to working memory, planning, inhibition, and flexibility, but the heterogeneous literature that largely was not designed to study the cerebellum makes it difficult to determine specific functions of the cerebellum or cerebellar regions. In addition, while it is clear that cerebellar insult in childhood affects executive function performance later in life, more work is needed to elucidate the mechanisms by which executive dysfunction occurs and its developmental course. The limitations of the current literature are discussed and potential directions for future research are provided.© 2021. Springer Science+Business Media, LLC, part of Springer Nature.